
A large group of asymmetrical chemical compounds can exist in two forms with the same molecular formula forming mirror images of one another, which means that they are chiral. Prof. Małgorzata Barańska from the JU Faculty of Chemistry will shed some light on this phenomenon studied by her research team, whose work has recently led to some major discoveries in this area.
What does chirality mean? In order to better understand this feature, let’s imagine a mirror. Putting a hypothetical molecule in front of it, we will get a reflection that precisely reproduces its structure. If the original molecule was placed next to its mirror image, we could observe one of two possible relations between them: molecule and its mirror image are either identical, which means that their structure is symmetric, or they don’t coincide with each other – the left side of the former looks like the right side of the latter and vice-versa. This difference constitutes the very essence of chirality. In the first case, the molecule remains visually the same, no matter whether we look at the original or the mirror image, whereas in the second case, the molecules are enantiomers, that is, optical “opposites” of one another.
A dextrorotatory steak
Chirality is a very common natural phenomenon. ‘In fact, the entire human body is built of enantiomers. The most common example are proteins, which play many roles in our bodies and the DNA, which contains the genetic code', explains Prof. Barańska. Actually, all amino acids, with the exception of the simplest one – glycine – are chiral compounds and, besides, are characterised by exactly the same L-configuration (the amino- group is located to the left of the main chain). Essential L-amino acids are necessary for the proper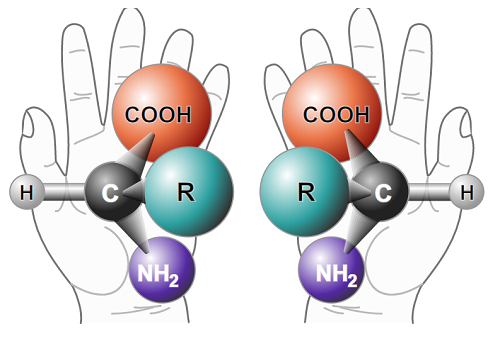 functioning of the body, which cannot create them on its own. Here comes the question whether mirror images of these substances would also be able to perform such functions. ‘We could, for instance, imagine or even synthesise a protein solely from D-amino acids (an amino-group located to the right of the main chain). It would look very similar to its “original” L-counterpart. The only and – as it turns out – very important difference, even though imperceptible to the naked eye, would be the fact that the structure of a D-protein would become a mirror image of an L-protein’, says Prof. Barańska. ‘If we managed to grow a piece of muscle (such cultivation is already possible) using only D-proteins and decided to make a dish out of such meat, it would turn out that it doesn’t contain the same nutrients as the “normal” one. D-amino acids wouldn’t be acquired from the food in the same way as those from the L group, because our body, which uses various enzymatic mechanisms (which are also chiral) wouldn’t “recognise” them’, says the researcher. The same applies to carbohydrates, almost all of which are of D-configuration when occurring naturally. What’s interesting, if we sweetened coffee with L-glucose, instead of D-glucose, the beverage would still be sweet, but would provide much less energy, as this version of the sugar is very poorly absorbed by the body, or even not absorbed at all.
functioning of the body, which cannot create them on its own. Here comes the question whether mirror images of these substances would also be able to perform such functions. ‘We could, for instance, imagine or even synthesise a protein solely from D-amino acids (an amino-group located to the right of the main chain). It would look very similar to its “original” L-counterpart. The only and – as it turns out – very important difference, even though imperceptible to the naked eye, would be the fact that the structure of a D-protein would become a mirror image of an L-protein’, says Prof. Barańska. ‘If we managed to grow a piece of muscle (such cultivation is already possible) using only D-proteins and decided to make a dish out of such meat, it would turn out that it doesn’t contain the same nutrients as the “normal” one. D-amino acids wouldn’t be acquired from the food in the same way as those from the L group, because our body, which uses various enzymatic mechanisms (which are also chiral) wouldn’t “recognise” them’, says the researcher. The same applies to carbohydrates, almost all of which are of D-configuration when occurring naturally. What’s interesting, if we sweetened coffee with L-glucose, instead of D-glucose, the beverage would still be sweet, but would provide much less energy, as this version of the sugar is very poorly absorbed by the body, or even not absorbed at all.
The way you smell is the way you... rotate
Another interesting example is thalidomide – a drug that was administered in the 1950s to pregnant women suffering from morning sickness. ‘Only its dextrorotatory form (R-thalidomide) had therapeutic properties, unlike its mirror image (S-thalidomide). The women were given pills with a 50/50 mixture of both enantiomers. After some time, it turned out that S-thalidomide led to the deformation of foetus’ limbs, especially when it was taken during the earliest weeks of pregnancy. About 12 thousand such cases were documented’, says Prof. Barańska. This brings up the question: why did they decide to use the mixture of both enantiomers if one of them didn’t have the therapeutic properties? ‘The decision was based on economic reasons. The synthesis resulting in racemic mixture [that is, consisting of both enantiomers] is simply much cheaper’, explains the researcher. This phenomenon is relatively common. For example, when we take one of the most popular painkillers, we provide our bodies with both emantiomers of ibuprofen, only one of which has painkilling properties (fortunately, the second one has no adverse effects). Enantiomers can also smell differently – for instance, the dextrorotatory enantiomer of carvone (an organic chemical compound) smells like mint, whereas the laevorotatory one smells like caraway.
A protector from chaos
What is the cause of chirality and why is it co common? An enantiomer usually has its chirality centre, such as a carbon atom, which is able to form bonds with four other atoms or groups of atoms. When there are minimal differences between them, e.g. in weight, there are only two possible ways of their spatial arrangement, which form mirror images of one another (see below).
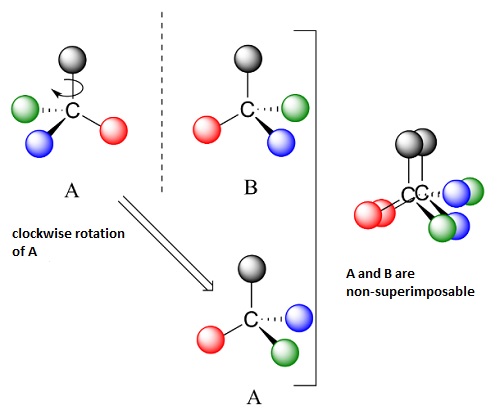 Does nature usually favour any of the two arrangements of molecules of a given substance? Why has it chosen, for instance, L-amino acids or D-carbohydrates and not the other way round? ‘We haven’t found any definite answer yet’, says Prof. Barańska. ‘What we know is the fact that at some point a random chiral factor probably influenced the way the world developed. The world definitely couldn’t be based on a system of proteins built from both kinds of amino acids’, adds the researcher. If DNA was built of both carbohydrate enantiomers (D and L), it would create too many options and possible evolutionary paths. ‘The more possibilities, the more errors, according to one of the hypotheses’, explains Prof. Barańska. So, the fact that proteins are built solely from L-amino acids has made the world much more orderly.
Does nature usually favour any of the two arrangements of molecules of a given substance? Why has it chosen, for instance, L-amino acids or D-carbohydrates and not the other way round? ‘We haven’t found any definite answer yet’, says Prof. Barańska. ‘What we know is the fact that at some point a random chiral factor probably influenced the way the world developed. The world definitely couldn’t be based on a system of proteins built from both kinds of amino acids’, adds the researcher. If DNA was built of both carbohydrate enantiomers (D and L), it would create too many options and possible evolutionary paths. ‘The more possibilities, the more errors, according to one of the hypotheses’, explains Prof. Barańska. So, the fact that proteins are built solely from L-amino acids has made the world much more orderly.
And what if the two enantiomers cross each other’s paths? ‘If there is a 50/50 mixture of both enantiomers, they neutralise one another’s optical activity, whereas in the case of a small but perceptible excess of one enantiomer, it’s possible to detect its characteristic signal. Yet, there is no significant change in terms of the substance’s properties. Sometimes, racemisation can occur - a process during which a set of molecules of one enantiomer is transformed into a racemic mixture of both of them.
A breakthrough coercion
Prof. Barańska’s team have found out that carotene crystals isolated from carrot root are chiral. What’s so special about it? The point is that the crystals contain 95% beta-carotene, which is an achiral compound, that is, identical with its mirror image. So how was it possible for such substance to be characterised with so intense optical activity? ‘In our study, we’ve shown that achiral molecules form crystals in an orderly manner. It turned out that a small amount of chiral compound caused other particles to become optically active too’, explains Prof. Barańska. The key factor was an almost identical structure of both substances, in accordance with the principle of similar interacting with similar. It’s much more difficult to “force” the achiral carotenoids to become chiral by, for instance, making them interact with an optically active carbohydrate of a much different structure. When the structure of a chiral carotenoind is only slightly different from an achiral one, interaction between is possible.
How did the scientists manage to induce chirality? According to Prof. Barańska, under certain conditions carotenoid particles cluster together into aggregates. This results from the simple fact that, from the energetic point of view it’s much better for them to be close to one another than to interact with a solvent (crystallisation process works likewise). The chiral aggregates are obviously created by chiral molecules, whereas the achiral aggregates – by achiral carotenoids. ‘However, it turns out that adding a small amount (5%) of a chiral carotenoid to an achiral one causes the resultant aggregate to display physical (spectroscopic) properties which we would expect from a fully chiral carotenoid. ‘And this is precisely what induction of chirality is about’, Prof. Barańska points out.
The other side of the universe
All this brings up another question, which takes us to the realm of science fiction – is it possible to create a universe that would be a mirror image of ours? ‘We could definitely imagine a universe that is built entirely from D-amino acids and L-carbohydrates. What’s interesting is that we wouldn’t even notice any difference. Even chirality observed in macro scale, exemplified, for instance, by snail shells, wouldn’t have any significant impact on our lives’, contends Prof. Barańska. In a world where L-amino acids and D-carbohydrates wouldn’t be absorbable and R-thalidomide would be harmful, instead of S-thalidomide, life would still go on. Of course, provided that we would find ourselves in this alternate reality from the very beginning, because, according to Prof. Barańska ‘in a would of proteins built of D-amino acids and L-sugars the inhabitants of our real planet wouldn’t have anything to eat, whereas the substances we encounter everyday, which now have positive or neutral impact on our health, could quickly annihilate us.’
The discoveries related to the induction of chirality have resulted from the collaborative effort of researchers from the Jagiellonian University Faculty of Chemistry, the Institute of Organic Chemistry at the Polish Academy of Sciences and the Institute of Plant Biology and Biotechnology at the University of Agriculture in Kraków.
Original text: www.nauka.uj.edu.pl





