The Journal of American Chemical Society has published a paper entitled Three-Dimensional Fully π-Conjugated Macrocycles: When 3D-Aromatic and When 2D-Aromatic-in-3D?, co-authored by Dr hab. Dariusz Szczepanik from the JU Department of Theoretical Chemistry. The paper was written in collaboration with researchers from the University of Girona, Uppsala University and Ghent University.
The concept of 3D aromaticity is particularly valuable in designing large and topologically complex three-dimensional chemical structures characterised by high thermodynamic stability and unique magnetic and transportation properties. Despite the rising popularity of 3D aromaticity amongst both theoretical and practical researchers, specific qualitative criteria necessary for the existence of this type of aromaticity were previously unknown.
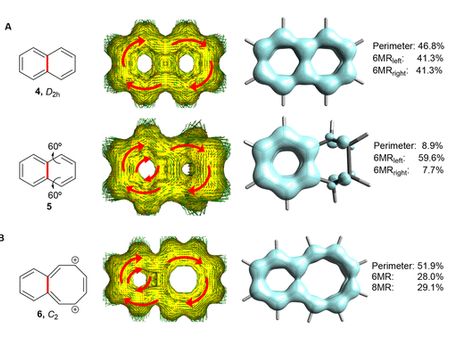
In the paper, Dr hab. Dariusz Szczepanik along with his collaborators presents a series of theoretical analyses using the latest computational methods, including the electron density of delocalised bond function (EDDB) invented at the JU Faculty of Chemistry, for the first time specifying the topological and structural-electron properties required for three-dimensional chemical structures to be 3D aromatic. Additionally, the researchers have shown that hitherto synthesised, three-dimensional particles suspected of 3D aromaticity are in fact 2D aromatic.
The research project was funded by the Polish National Science Centre within the framework of the Sonata Bis 11 call under the supervision of Dr hab. Dariusz Szczepanik. The full version of the paper is available free of charge on the ACS Publications website.