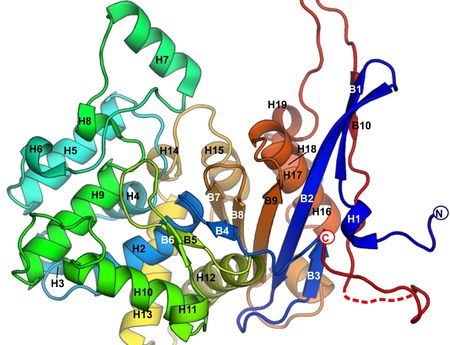
On 18 November, the journal Nature Communications published a paper entitled Crystal structures of the elusive Rhizobium etli L-asparaginase reveal a peculiar active site. The publication is a joint effort by the Adam Mickiewicz University in Poznań, Polish Academy of Sciences Institute of Chemistry and the Jagiellonian University.
The paper describes the properties of a newly discovered enzyme called ReAV which breaks down asparagine into acid and ammonia. Enzymes that perform that function, called asparaginases, are extremely important for medical reasons, since they can prove to be very effective at treating leukaemia in children. The enzyme described in the paper does not meet all the requirements necessary to produce an effective drug, but studying it is a crucial step in the right direction. The ReAV enzyme's structure has completely surprised the research team, which is not insignificant considering their expertise in the field. As the research into ReAV's structure has been going on for nearly 20 years, the scale of the achievement is impressive. The study was funded by as grant from the National Science Centre.
The paper was written by a research team working under the supervision of Prof. Mariusz Jaskólski from the Polish Academy of Sciences Department of Crystallography. The team also includes Dr Joanna Loch and Anna Ściuk from the JU Department of Crystal Chemistry and Crystal Physics.





